Difference between revisions of "Aging and Storage"
| (37 intermediate revisions by the same user not shown) | |||
| Line 16: | Line 16: | ||
Bottle conditioning is the process and changes that take a beer at packaging time to beer that is ready to drink. This can include the development of carbonation, microbial growth, development and reprocessing of off flavors, 'bottle shock' and other changes. Bottle conditioning, at least for the initial period where carbonation is generated, is typically carried out at warmer temperatures than extended aging after the conditioning is done. | Bottle conditioning is the process and changes that take a beer at packaging time to beer that is ready to drink. This can include the development of carbonation, microbial growth, development and reprocessing of off flavors, 'bottle shock' and other changes. Bottle conditioning, at least for the initial period where carbonation is generated, is typically carried out at warmer temperatures than extended aging after the conditioning is done. | ||
| + | |||
| + | See also: | ||
| + | * [https://encyclopedia.pub/item/revision/d2232da8945e383a5a0cdf189b38a2d8 "Bottle Conditioning," Topic Review by Kateřina Štulíková and Pavel Dostálek, Scholarly Community Encyclopedia.] | ||
| + | * [[Packaging#Oxygen_exposure|Packaging and Oxygen Exposure.]] | ||
| + | * [https://www.facebook.com/groups/MilkTheFunk/permalink/2282287018466166/ Justin Amaral's can conditioning project with DO tracking for canned conditioned beers on MTF.] | ||
==Techniques of Cellaring== | ==Techniques of Cellaring== | ||
Cellaring, or extended age in the bottle once the beer is ready to drink, is common for many mixed fermentation beers. Cellaring is typically carried out at cooler temperatures. | Cellaring, or extended age in the bottle once the beer is ready to drink, is common for many mixed fermentation beers. Cellaring is typically carried out at cooler temperatures. | ||
| − | ===Bottles vs Kegs=== | + | ===Bottles vs Kegs vs Cans vs PET=== |
| + | Chemical changes over time can be different as the beer ages. The packaging type can have a significant impact on how the beer ages. One study on lager found that PET bottles had the greatest variation in chemical changes over a 6 month period of time at ~20°C compared to glass bottles, kegs, and cans. In particular, diacetyl was higher in PET bottles as the beer aged. This was attributed to the PET bottles being more permeable to oxygen because of oxidation of acetoin and 2,3-butanediol to form diacetyl, or the oxidative decarboxylation of alpha-acetolactate, a precursor to diacetyl. Cans showed the least formation of diacetyl, while kegs and bottles displayed moderate increases in diacetyl over time compared to the PET bottles. Acetaldehyde was also highest in the PET bottles. DMS had a high spike during the first month of storage, but by the end of 6 months, the PET bottles had less DMS than the other storage types and glass bottles had the most. Other compounds had less significant differences between package type (ethyl acetate, propanol, isobutanol, isoamyl alcohol, higher alcohols, and esters) <ref>[https://www.sciencedirect.com/science/article/pii/S2214289420300089 The influence of packaging material on volatile compounds of pale lager beer. Goran Gagula, Kristina Mastanjević, Krešimir Mastanjević, Vinko Krstanović, Daniela Horvat, Damir Magdić. 2020.]</ref>. | ||
===Corks vs Caps=== | ===Corks vs Caps=== | ||
| Line 26: | Line 32: | ||
====CO<sup>2</sup> Loss Over Time==== | ====CO<sup>2</sup> Loss Over Time==== | ||
| − | Young finished champagne and sparkling wines produced according to the ''méthode traditionnelle'' process, which involves carbonating the champagne with sugar for 15 months and then disgorging them and corking them, begin with a CO<sup>2</sup> concentration of around 11-12 g/L (~6 volumes), while sparkling wines that are 5 years old and 10 years old have been found to have a much lower concentration of CO<sup>2</sup> at around 6-8 g/L (~3-4 volumes) <ref>[https://www.sciencedirect.com/science/article/pii/S000326700901349X?via%3Dihub CO2 volume fluxes outgassing from champagne glasses: The impact of champagne | + | Young finished champagne and sparkling wines produced according to the ''méthode traditionnelle'' process, which involves carbonating the champagne with sugar for 15 months and then disgorging them and corking them, begin with a CO<sup>2</sup> concentration of around 11-12 g/L (~6 volumes), while sparkling wines that are 5 years old and 10 years old have been found to have a much lower concentration of CO<sup>2</sup> at around 6-8 g/L (~3-4 volumes) <ref>[https://www.sciencedirect.com/science/article/pii/S000326700901349X?via%3Dihub CO2 volume fluxes outgassing from champagne glasses: The impact of champagne aging. Gérard Liger-Belair, Sandra Villaume, Clara Cilindre, Philippe Jeandet. 2010.]</ref><ref name="Liger-Belair_2011">[https://pubs.acs.org/doi/abs/10.1021/jf104675s Losses of Dissolved CO2 Through the Cork Stopper during Champagne Aging: Toward a Multiparameter Modeling. Gérard Liger-Belair and Sandra Villaume. 2011.]</ref>. The gradual loss of carbonation in sparkling wines has been attributed to the porous nature of corks allowing for the slow diffusion of gasses through them, which is highly variable based on the density of the cork <ref>[https://www.ncbi.nlm.nih.gov/pubmed/19215133 Kinetics of CO(2) fluxes outgassing from champagne glasses in tasting conditions: the role of temperature. Liger-Belair G1, Villaume S, Cilindre C, Jeandet P. 2009.]</ref><ref>[https://www.sciencedirect.com/science/article/pii/S0003267009013981?via%3Dihub#tbl1 Foaming properties of various Champagne wines depending on several parameters: Grape variety, aging, protein and CO2 content. Clara Cilindrea, Gérard Liger-Belair, Sandra Villaume, Philippe Jeandet, Richard Marchal. 2010.]</ref>, as well as the interface between the cork and the neck of the bottle <ref name="Liger-Belair_2011" />. An interesting observation is that there wasn't a large difference in carbonation loss between 5-year-old sparkling wines and 10-year-old sparkling wines, indicating that the loss of carbonation could greatly slow down once the liquid inside reaches around 3-4 volumes of CO<sup>2</sup>. |
The construction of the cork itself is a variable that makes it difficult to predict the exact rate of CO<sup>2</sup> loss. Corks are composed of two distinct parts: the mushroom of the cork is made up of agglomerated cork small granules, while the foot of the cork is made up of two large cork slices. This lower part is made up of several [https://en.wikipedia.org/wiki/Lenticel lenticels], which are parts of the plant that allow gasses to flow in and out of the plant. These lenticels vary from cork to cork. Nevertheless, a model has been proposed by Liger-Belair et al. that estimates the amount of CO<sup>2</sup> loss over time. In this model, two other variables have been identified as playing a large role in how much CO<sup>2</sup> is lost: storage temperature and bottle size. The warmer the storage temperature, the faster the rate is of losing of CO<sup>2</sup>, and the larger the bottle volume the slower the rate is of losing CO<sup>2</sup>. Below are some estimated CO<sup>2</sup> levels based on the Liger-Belair model in g/L and then converted to volumes in parenthesis at various points in time. The first table shows the estimated amount of CO<sup>2</sup> loss when stored at three different temperatures (4 °C, 12 °C, and 20 °C). The second table shows the estimated amount of CO<sup>2</sup> loss in different sized bottles (1.5 L, 750 mL, and 350 mL) when stored at 12 °C <ref name="Liger-Belair_2011" />: | The construction of the cork itself is a variable that makes it difficult to predict the exact rate of CO<sup>2</sup> loss. Corks are composed of two distinct parts: the mushroom of the cork is made up of agglomerated cork small granules, while the foot of the cork is made up of two large cork slices. This lower part is made up of several [https://en.wikipedia.org/wiki/Lenticel lenticels], which are parts of the plant that allow gasses to flow in and out of the plant. These lenticels vary from cork to cork. Nevertheless, a model has been proposed by Liger-Belair et al. that estimates the amount of CO<sup>2</sup> loss over time. In this model, two other variables have been identified as playing a large role in how much CO<sup>2</sup> is lost: storage temperature and bottle size. The warmer the storage temperature, the faster the rate is of losing of CO<sup>2</sup>, and the larger the bottle volume the slower the rate is of losing CO<sup>2</sup>. Below are some estimated CO<sup>2</sup> levels based on the Liger-Belair model in g/L and then converted to volumes in parenthesis at various points in time. The first table shows the estimated amount of CO<sup>2</sup> loss when stored at three different temperatures (4 °C, 12 °C, and 20 °C). The second table shows the estimated amount of CO<sup>2</sup> loss in different sized bottles (1.5 L, 750 mL, and 350 mL) when stored at 12 °C <ref name="Liger-Belair_2011" />: | ||
| Line 67: | Line 73: | ||
* [https://www.facebook.com/groups/MilkTheFunk/permalink/1779034952124711/?comment_id=1779340158760857&reply_comment_id=1779452025416337&comment_tracking=%7B%22tn%22%3A%22R4%22%7D Blake Tyers from Creature Comforts] reported no statistical significance been horizontal and vertical storage, however those that did identify a difference correctly noticed harsher flavors and "more edges" in the bottles stored upright. | * [https://www.facebook.com/groups/MilkTheFunk/permalink/1779034952124711/?comment_id=1779340158760857&reply_comment_id=1779452025416337&comment_tracking=%7B%22tn%22%3A%22R4%22%7D Blake Tyers from Creature Comforts] reported no statistical significance been horizontal and vertical storage, however those that did identify a difference correctly noticed harsher flavors and "more edges" in the bottles stored upright. | ||
* Ryan Fields reported not seeing a difference between horizontal versus vertical storage, however, they only tested this one time. He still prefers to age bottles vertically in cages because this requires less space than aging in stacked boxes, and aging in boxes can insulate from proper airflow and temperature <ref name="fields_goodwin" /> (~29:45). | * Ryan Fields reported not seeing a difference between horizontal versus vertical storage, however, they only tested this one time. He still prefers to age bottles vertically in cages because this requires less space than aging in stacked boxes, and aging in boxes can insulate from proper airflow and temperature <ref name="fields_goodwin" /> (~29:45). | ||
| + | * [https://www.facebook.com/groups/MilkTheFunk/permalink/2649241528437378/ Alex Levy reported off-flavors in MTF] when storing bottles that were capped (no cork) horizontally but not vertically. | ||
* For equipment and methodology of using wire cages to store horizontally, see [[Packaging#Wire_Storage_Containers|Wire Storage Containers]]. | * For equipment and methodology of using wire cages to store horizontally, see [[Packaging#Wire_Storage_Containers|Wire Storage Containers]]. | ||
| Line 77: | Line 84: | ||
[http://www.sciencedirect.com/science/article/pii/S0740002014002548 Spitaels et al., 2015 microbes in bottles of gueuze] | [http://www.sciencedirect.com/science/article/pii/S0740002014002548 Spitaels et al., 2015 microbes in bottles of gueuze] | ||
| + | |||
| + | [https://www.facebook.com/groups/MilkTheFunk/posts/7284659538228864/ Anecdotal evidence that ''Brettanomyces'' can reverse discoloration from enzymatic browning.] | ||
See also [[Commercial Sour Beer Dregs Inoculation]]. | See also [[Commercial Sour Beer Dregs Inoculation]]. | ||
| Line 100: | Line 109: | ||
* Sulfur compounds: dimethyl trisulfide (production enhanced by low pH), 3-Methyl-3-mercaptobutylformate. | * Sulfur compounds: dimethyl trisulfide (production enhanced by low pH), 3-Methyl-3-mercaptobutylformate. | ||
| − | ===General Effects of | + | ===General Effects of Oxidation=== |
| − | + | Oxidation, also known as a redox reaction, is the chemical process of atoms losing electrons to other atoms. The atom that loses an electron is called the "oxidizer", and the atom that gains the electron is called the "reducer". Despite the name of this process being called "oxidation" and oxygen often being the reducer, oxygen is not required since other chemicals can serve this purpose. Oxidation can occur slowly (e.g. metal rusting) or quickly (e.g. fire), and applies to a large range of simple and complex processes <ref>[https://en.wikipedia.org/wiki/Redox Wikipedia. "Redox". Retrieved 09/03/2017.]</ref>. In beer, oxidation takes the form of carbon-based molecules or metal ions losing electrons to either oxygen molecules or free radicals. Oxygen itself in its ground state is not particularly reactive in beer, however, oxygen in beer reacts with transition metal ions found in beer such as copper, iron, and manganese (these include two types of redox reactions called the Fenton, and Haber-Weiss reactions) to form "reactive oxygen species" (ROS) which then react with other compounds in the beer to cause staling <ref name="Barnette_2018_Masters">[http://scholar.google.com/scholar_url?url=https://ir.library.oregonstate.edu/downloads/dv140033b&hl=en&sa=X&d=799257176923188618&scisig=AAGBfm23Uy0QqVLXJEUSylw-LILNTHHd7Q&nossl=1&oi=scholaralrt&hist=CYJIrnMAAAAJ:10241589793194662084:AAGBfm17pAuQUDgk8QVeubsITC7flr3nZQ Evaluating the Impact of Dissolved Oxygen and Aging on Dry-Hopped Aroma Stability in Beer. Bradley M. Barnette. Masters Thesis in Food Science and technology, Oregon State University. 2018.]</ref>. Oxidation reactions increase the amount of off-flavor compounds, as well as dulls the aroma of beer. Examples of off-flavors produced by these redox processes include aldehydes and ketones (e.g. acetaldehyde), trans-2-nonenol, and diacetyl. Beer also darkens in color when exposed to oxygen, probably through an oxidation process known as [https://en.wikipedia.org/wiki/Food_browning enzymatic browning]. Brewers yeast and ''Brettanomyces'' are great scavengers of oxygen, and adding fresh yeast and sugar at packaging can help reduce dissolved oxygen in the package, and even reverse some effects of oxidation <ref>[http://pubs.acs.org/doi/abs/10.1021/jf9037387 Decrease of Aged Beer Aroma by the Reducing Activity of Brewing Yeast. Daan Saison, David P. De Schutter, Nele Vanbeneden, Luk Daenen, Filip Delvaux and Freddy R. Delvaux. 2010.]</ref><ref name="hall_mitchell" /><ref name="Barnette_2018_Masters" />. | |
| − | 2018.]</ref>. Oxidation | ||
| − | In general, the best practice is to limit oxygen at packaging time, although brewers have had success packaging beers with living ''Brettanomyces'' without purging the bottles with CO<sup>2</sup>. Other compounds can serve as anti-oxidants in beer. For example, sulfates are converted into sulfites by yeast, and sulfites postpone the formation of free radicals. Lower-weight polyphenols, which originate from malt (70-80%) and hops (20-30%), are thought to be free radical scavengers and anti-oxidants, however other polyphenols have been identified as pro-oxidants and the effectiveness of antioxidant activity in general for polyphenols is debatable in the scientific literature (although their impact in the mash and boil has been established as positive). Maillard reactions from malting/roasting and wort boiling also create anti-oxidants, and in general the darker the roasting the more anti-oxidant the malts will be <ref name="Vanderhaegen_2006" />, although compounds in kilned malts, hypothesized to be the proanthocyanidins and flavonols derived from Maillard reactions, have been found to be a source for oxidation and beer staling. Alpha acids and iso-alpha acids have been shown to react with transition metal ions (iron), thus reducing the impact of the oxidation of iron ions <ref name="Barnette_2018_Masters" />. Lactic acid and lactic acid fermentation are thought to also help serve as anti-oxidants, although this has not be studied in sour beer <ref>[https://www.ncbi.nlm.nih.gov/pubmed/10904049 Free radical scavenging and antioxidant effects of lactate ion: an in vitro study. Groussard C, Morel I, Chevanne M, Monnier M, Cillard J, Delamarche A. 1985.]</ref><ref>[https://www.sciencedirect.com/science/article/pii/S0740002011000530 Effect of lactic acid fermentation on antioxidant, texture, color and sensory properties of red and green smoothies. Raffaella Di Cagno, Giovanna Minervini, Carlo G. Rizzello, Maria De Angelis, Marco Gobbetti. 2011.]</ref>. | + | In general, the best practice is to limit dissolved oxygen (DO) levels to 30-60 ppb (or 40-150 ppb [https://tapintohach.com/2014/03/18/dissolved-oxygen-in-beer-how-it-compares-to-total-package-oxygen/ total package oxygen (TPO)]) <ref>[https://www.hach.com/cms-portals/hach_com/cms/documents/pdf/LIT2149-how-to-measure-DO-in-a-brewery.pdf "HOW TO MEASURE DISSOLVED OXYGEN IN THE BREWERY". Hach Company pamphlet. Retrieved 10/23/2018.]</ref>, much of which is picked up at packaging time, although brewers have had success packaging beers with living ''Brettanomyces'' without purging the bottles with CO<sup>2</sup>. Dissolved oxygen should be measured with a dissolved oxygen sensor/meter during production and immediately at packaging; Bradley Barnette's Masters thesis showed that beers that were oxygenated at packaging to test the effect on dry hopping at 200 ppb had similar DO levels to beers oxygenated at 50 ppb after two weeks of storage, which lead to the hypothesis that oxygen is consumed via oxidation reactions over time during the storage of beer <ref name="Barnette_2018_Masters" />. Other compounds can serve as anti-oxidants in beer. For example, sulfates are converted into sulfites by yeast, and sulfites postpone the formation of free radicals. Lower-weight polyphenols, which originate from malt (70-80%) and hops (20-30%), are thought to be free radical scavengers and anti-oxidants, however other polyphenols have been identified as pro-oxidants and the effectiveness of antioxidant activity in general for polyphenols is debatable in the scientific literature (although their impact in the mash and boil has been established as positive). Maillard reactions from malting/roasting and wort boiling also create anti-oxidants, and in general the darker the roasting the more anti-oxidant the malts will be <ref name="Vanderhaegen_2006" />, although compounds in kilned malts, hypothesized to be the proanthocyanidins and flavonols derived from Maillard reactions, have been found to be a source for oxidation and beer staling. Alpha acids and iso-alpha acids have been shown to react with transition metal ions (iron), thus reducing the impact of the oxidation of iron ions <ref name="Barnette_2018_Masters" />. Lactic acid and lactic acid fermentation are thought to also help serve as anti-oxidants, although this has not be studied in sour beer <ref>[https://www.ncbi.nlm.nih.gov/pubmed/10904049 Free radical scavenging and antioxidant effects of lactate ion: an in vitro study. Groussard C, Morel I, Chevanne M, Monnier M, Cillard J, Delamarche A. 1985.]</ref><ref>[https://www.sciencedirect.com/science/article/pii/S0740002011000530 Effect of lactic acid fermentation on antioxidant, texture, color and sensory properties of red and green smoothies. Raffaella Di Cagno, Giovanna Minervini, Carlo G. Rizzello, Maria De Angelis, Marco Gobbetti. 2011.]</ref>. |
| + | |||
| + | Oxygen has a large and negative impact on highly hopped beers. Dry hopping also serves as a greater risk of oxidation in beer. Oxygen can enter the beer during the dry hopping process. It's also been shown that iron ions increase from hop additions, which react with oxygen and cause oxidation. Higher DO can slightly increase the rate of IBU degradation. Interestingly, during two weeks of storage it was shown by Barnette that hop compounds such as monoterpenes did not decline even though flavor analysis reported decreased hoppy, fruity, and citrus character, suggesting that this is caused by the production of other oxidized compounds in the beer rather than a decrease hop-derived compounds <ref name="Barnette_2018_Masters" />. | ||
See also: | See also: | ||
| + | * [https://www.morebeer.com/articles/oxidation_in_beer "Controlling Beer Oxidation" by George Fix.] | ||
* [https://jp.hach.com/asset-get.download.jsa?id=50544340479 Industry standards of dissolved oxygen levels in beer throughout the brewing process, by Hach.] | * [https://jp.hach.com/asset-get.download.jsa?id=50544340479 Industry standards of dissolved oxygen levels in beer throughout the brewing process, by Hach.] | ||
| + | * [https://www.facebook.com/groups/MilkTheFunk/permalink/3835629766465209/ MTF thread on anecdotal accounts of ''Brettanomyces'' affecting oxidation character in beer.] | ||
| + | * [https://suigenerisbrewing.com/index.php/2022/03/12/metabisulfite-7-year-experiment/ Dr. Bryan Heit explanations chemical oxidation pathways, and the use of metabisulfite to limit oxidation in packaging.] | ||
===General Effects of Temperature=== | ===General Effects of Temperature=== | ||
| − | The temperature at which beer is stored has also has a major impact on how beer ages. The effect that temperature has on a given reaction depends on the type of reaction; not all reactions are increased at the same rate. For example, it has been reported that beer stored at 25°C tends to develop more caramel flavor, while the same beer stored at 30°C develops more cardboard flavor <ref name="Vanderhaegen_2006" />. Bamforth and Lentini proposed a simplified version of the Arrhenius model known as [https://web.calpoly.edu/~bio/EPL/pdfs/SampleLectureBIO162.pdf | + | The temperature at which beer is stored has also has a major impact on how beer ages. The effect that temperature has on a given reaction depends on the type of reaction; not all reactions are increased at the same rate. For example, it has been reported that beer stored at 25°C tends to develop more caramel flavor, while the same beer stored at 30°C develops more cardboard flavor <ref name="Vanderhaegen_2006" />. Bamforth and Lentini proposed a simplified version of the Arrhenius model known as [https://web.calpoly.edu/~bio/EPL/pdfs/SampleLectureBIO162.pdf Q<sub>10</sub>] to generalize the rate of chemical reactions in beer based on temperature. Q<sub>10</sub> is a measure of the temperature sensitivity of chemical and enzymatic reactions due to an increase in temperature by 10°C. The equation is expressed as a ratio: Q<sub>10</sub> = Reaction Time + 10°C / Reaction Time. Bamforth recommends that for beer, Q<sub>10</sub> will be 2 or 3 (most chemical reactions fall in this range). For example, assuming a Q<sub>10</sub> of 3, storing beer at 30°C for 2 weeks is equivalent to 6 weeks at 20°C, 18 weeks at 10°C, and 54 weeks at 1°C. Assuming a Q<sub>10</sub> of 2, storing beer at 30°C for 2 weeks is equivalent to 4 weeks at 20°C, 8 weeks at 10°C, and 16 weeks at 1°C <ref>[https://www.sciencedirect.com/science/article/pii/B9780126692013000038 Charles Bamforth and Aldo Lentini. Beer: A Quality Perspective. 2009. Pgs 85-109.]</ref><ref name="Barnette_2018_Masters" /><ref>[https://web.calpoly.edu/~bio/EPL/pdfs/SampleLectureBIO162.pdf Temperature Regulation PowerPoint. Cal Poly. Environmental Proteomics Laboratory.]</ref>. Barnette's Masters thesis found that warmer temperatures had a greater negative impact on hop flavor and aroma than high levels of dissolved oxygen over a two week storage period in dry hopped beers <ref name="Barnette_2018_Masters" />. |
===Haze=== | ===Haze=== | ||
| Line 130: | Line 143: | ||
http://www.sciencedirect.com/science/article/pii/S0740002014002548 | http://www.sciencedirect.com/science/article/pii/S0740002014002548 | ||
| − | Some volatile esters that give the beer a fruity flavor, such as isoamyl acetate that gives the beer a banana flavor, can decrease to levels below their flavor thresholds over time in packaged beer. Other volatile esters such as ethyl 3-methyl-butyrate (contributes a wine-like character), ethyl 2-methyl-butyrate (contributes a wine-like character), ethyl 2-methyl-propionate, ethyl nicotinate, diethyl succinate, ethyl lactate, ethyl phenylacetate, ethyl formate, ethyl furoate and ethyl cinnamate (fruity, sweet character) are formed during beer aging. Lactones, which are cyclic esters, such as the peach-like hexalactone and nonalactone tend to increase during beer storage and have a significant impact on the flavor of aged beer <ref name="Vanderhaegen_2006" />. | + | Some volatile esters that give the beer a fruity flavor, such as isoamyl acetate that gives the beer a banana flavor, can decrease to levels below their flavor thresholds over time in packaged beer. Other volatile esters such as ethyl 3-methyl-butyrate (derived from oxidized alpha and beta acids in hops and contributes a wine-like character <ref name="williams_wagner_1979">[https://www.asbcnet.org/publications/journal/vol/Abstracts/37-03.htm Contribution of Hop Bitter Substances to Beer Staling Mechanisms. Williams and Wagner 1979.]</ref>), ethyl 2-methyl-butyrate (derived from oxidized alpha and beta acids in hops and contributes a wine-like character <ref name="williams_wagner_1979" />), ethyl 2-methyl-propionate, ethyl nicotinate, diethyl succinate, ethyl lactate, ethyl phenylacetate, ethyl formate, ethyl furoate and ethyl cinnamate (fruity, sweet character) are formed during beer aging. Lactones, which are cyclic esters, such as the peach-like hexalactone and nonalactone tend to increase during beer storage and have a significant impact on the flavor of aged beer <ref name="Vanderhaegen_2006" />. See also [[Hops#Esters|Esters From Hops]]. |
While most ester formation and hydrolysis (breakdown) in beer during aging are mostly acid-catalysed, some of this activity is due to esterase enzymes. It has been shown that some esterases are released by yeast autolysis during beer aging. This release of esterase enzymes is strain dependent, with ale yeast having more potential for this activity than lager yeast. The enzyme is most active at 15-20°C and is destroyed by pasteurization <ref name="Vanderhaegen_2006" />. | While most ester formation and hydrolysis (breakdown) in beer during aging are mostly acid-catalysed, some of this activity is due to esterase enzymes. It has been shown that some esterases are released by yeast autolysis during beer aging. This release of esterase enzymes is strain dependent, with ale yeast having more potential for this activity than lager yeast. The enzyme is most active at 15-20°C and is destroyed by pasteurization <ref name="Vanderhaegen_2006" />. | ||
| Line 155: | Line 168: | ||
[https://en.wikipedia.org/wiki/Polyphenol Polyphenols] are a large group of organic chemicals characterized by many phenol structures combined. Subclasses of polyphenols include tannic acid, tannins, and flavonoids <ref name="wikipedia_polyphenols" /><ref>[https://en.wikipedia.org/wiki/Flavonoid Flavonoid. Wikipedia website. Retrieved 05/02/2017.]</ref>. | [https://en.wikipedia.org/wiki/Polyphenol Polyphenols] are a large group of organic chemicals characterized by many phenol structures combined. Subclasses of polyphenols include tannic acid, tannins, and flavonoids <ref name="wikipedia_polyphenols" /><ref>[https://en.wikipedia.org/wiki/Flavonoid Flavonoid. Wikipedia website. Retrieved 05/02/2017.]</ref>. | ||
| − | Polyphenols have an ambiguous role in the aging of beer. Flavonoids (for example catechin, which comes from hops and is a major source of polyphenols in beer), are antioxidants and protect more sensitive compounds such as isohumulones from oxidation by scavenging free radicals and binding with oxidative metals (iron, for example). However, they themselves can also be oxidized over time to possibly create off-flavors. In addition to their own oxidation, [https://en.wikipedia.org/wiki/Hydroxyl_radical hydroxyl radicals] that cause oxidation also react highly with ethanol, and therefore a portion may not react with polyphenols. After a lag period of 5 weeks in the bottle, it was found that levels of tannins actually increase. This is thought to be caused by smaller flavonoids reacting with acetaldehyde. Polyphenols were also oxidized into [https://en.wikipedia.org/wiki/Quinone quinones], which are a stepping stone in the reaction that causes [https://en.wikipedia.org/wiki/Food_browning oxidative food browning]. The use of polyphenols during mashing and boiling has been shown to decrease trans-2-nonenal (cardboard flavor) and trans-2-nonenal that is protected from fermentation by being bound to proteins (see [[Aging_and_Storage#Tannic_Acid|Tannic Acid]] below). In two studies, there appeared to be no significant effect on free radical formation by polyphenols, probably due to the fact that they readily react with ethanol <ref name="Callemien_2010" />. | + | Polyphenols have an ambiguous role in the aging of beer. Flavonoids (for example catechin, which comes from hops and is a major source of polyphenols in beer), are antioxidants and protect more sensitive compounds such as isohumulones from oxidation by scavenging free radicals and binding with oxidative metals (iron, for example). However, they themselves can also be oxidized over time to possibly create off-flavors. In addition to their own oxidation, [https://en.wikipedia.org/wiki/Hydroxyl_radical hydroxyl radicals] that cause oxidation also react highly with ethanol, and therefore a portion may not react with polyphenols. After a lag period of 5 weeks in the bottle, it was found that levels of tannins actually increase. This is thought to be caused by smaller flavonoids reacting with acetaldehyde. Polyphenols were also oxidized into [https://en.wikipedia.org/wiki/Quinone quinones], which are a stepping stone in the reaction that causes [https://en.wikipedia.org/wiki/Food_browning oxidative food browning] (this reaction increases in the presence of acids <ref>[https://en.wikipedia.org/wiki/Quinone#Reduction "Quinone". Wikipedia. Retrieved 12/23/2023.]</ref>). The use of polyphenols during mashing and boiling has been shown to decrease trans-2-nonenal (cardboard flavor) and trans-2-nonenal that is protected from fermentation by being bound to proteins (see [[Aging_and_Storage#Tannic_Acid|Tannic Acid]] below). In two studies, there appeared to be no significant effect on free radical formation by polyphenols, probably due to the fact that they readily react with ethanol <ref name="Callemien_2010" />. |
Higher temperatures increase the rate of oxidized polyphenols. In one study on aged lagers, 6.5% of the polyphenols were oxidized after 5 days at 40°C/104°F, but only 0.6% of the polyphenols were oxidized after 9 months at 20°C/68°F <ref name="Callemien_2010" />. | Higher temperatures increase the rate of oxidized polyphenols. In one study on aged lagers, 6.5% of the polyphenols were oxidized after 5 days at 40°C/104°F, but only 0.6% of the polyphenols were oxidized after 9 months at 20°C/68°F <ref name="Callemien_2010" />. | ||
| Line 176: | Line 189: | ||
===Hop Compounds=== | ===Hop Compounds=== | ||
| + | |||
| + | - https://www.tandfonline.com/doi/full/10.1080/03610470.2019.1705037?needAccess=true | ||
| + | - https://www.tandfonline.com/doi/abs/10.1080/03610470.2020.1843898 | ||
====IBU Degradation==== | ====IBU Degradation==== | ||
| Line 181: | Line 197: | ||
====Lightstruck==== | ====Lightstruck==== | ||
| − | https://beersensoryscience.wordpress.com/2011/03/17/lightstruck/ | + | - https://beersensoryscience.wordpress.com/2011/03/17/lightstruck/ |
| − | http://www.scielo.br/scielo.php?pid=S0100-40422000000100019&script=sci_arttext&tlng=es | + | - http://www.scielo.br/scielo.php?pid=S0100-40422000000100019&script=sci_arttext&tlng=es |
| − | http://onlinelibrary.wiley.com/doi/10.1002/j.2050-0416.2002.tb00568.x/abstract | + | - http://onlinelibrary.wiley.com/doi/10.1002/j.2050-0416.2002.tb00568.x/abstract |
| − | http://www.professorbeer.com/articles/skunked_beer.html | + | - http://www.professorbeer.com/articles/skunked_beer.html |
| − | Oxidized alpha acids (humulinones) will skunk if exposed to UV light <ref>[https://www.homebrewersassociation.org/how-to-brew/resources/conference-seminars Dr. Patricia Aron. "Bitterness and the IBU: What’s It All About?" HomebrewCon 2017 Presentation. ~32 mins in. Retrieved 09/05/2017.]</ref>. | + | - https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/1521-3765%2820011105%297%3A21%3C4553%3A%3AAID-CHEM4553%3E3.0.CO%3B2-0 |
| + | |||
| + | - https://pubs.rsc.org/en/content/articlelanding/2004/pp/b316210a/unauth#!divAbstract | ||
| + | |||
| + | Iso-alpha acids will skunk if exposed to UV light. Oxidized alpha acids (humulinones) will also skunk if exposed to UV light <ref>[https://www.homebrewersassociation.org/how-to-brew/resources/conference-seminars Dr. Patricia Aron. "Bitterness and the IBU: What’s It All About?" HomebrewCon 2017 Presentation. ~32 mins in. Retrieved 09/05/2017.]</ref>. This compound is known as 3-methyl-2-butene-1-thiol (3MBT). Brown bottles filter most UV light, while green bottles only filter a portion of UV light. See [https://beerandbrewing.com/dictionary/eIXf22Zwnt/ "Lightstruck", Craft beer and Brewing Magazine website]. | ||
| + | |||
| + | * [https://www.youtube.com/watch?app=desktop&v=W4vJ9DhoLp4&t=774s Olivier Dedeycker explains why Saison Dupont is packaged in brown bottles for the US market and green bottles for the European market.] | ||
====Damascenone==== | ====Damascenone==== | ||
| − | The | + | The ketone beta-damascenone (rhubarb, red fruits, stewed apples; threshold of 25 ppb <ref name="hall_mitchell" />) is thought to be formed from the oxidation of hop oils, although it has also been found in unhopped aged beers. Potential precursors are allene triols and acetylene diols formed from the degradation of the carotenoid neoxanthin. It was found to increase greatly in aged beer that was at a pH of 3 or 4.2 versus a higher pH. This was attributed to the acidic hydrolysis of glycosides. The release of flavor compounds from glycosides could be present in acidic beers that are aged on fruit or herbs <ref>[https://pubs.acs.org/doi/full/10.1021/jf020085i Investigation of the β-Damascenone Level in Fresh and Aged Commercial Beers. Fabienne Chevance, Christine Guyot-Declerck, Jérôme Dupont, and Sonia Collin. 2002. DOI: 10.1021/jf020085i.]</ref><ref name="Vanderhaegen_2006" />. See [[Glycosides#Acidic_Hydrolysis|Glycosides]] for more information on acidic hydrolysis of glycosides. Damascenone is also found in grapes and is a major flavor component of bourbon <ref name="hall_mitchell" />. |
===Other Flavor and Non-flavor Compounds=== | ===Other Flavor and Non-flavor Compounds=== | ||
| Line 202: | Line 224: | ||
Products of Maillard reactions, which include a diverse range of reactions, have also been found in beer, although research in this area is limited. Some Maillard compounds found in aging beer remain under taste threshold, for example, furfural and 5-hydroxymethyl furfural. It is hypothesized that a wide range of unknown Maillard reactions and their intermediates might play a role in the aging of beer. In particular, the bready, sweet, caramel and wine-like character of stale beer might be due to Maillard reactions <ref name="Vanderhaegen_2006" />. | Products of Maillard reactions, which include a diverse range of reactions, have also been found in beer, although research in this area is limited. Some Maillard compounds found in aging beer remain under taste threshold, for example, furfural and 5-hydroxymethyl furfural. It is hypothesized that a wide range of unknown Maillard reactions and their intermediates might play a role in the aging of beer. In particular, the bready, sweet, caramel and wine-like character of stale beer might be due to Maillard reactions <ref name="Vanderhaegen_2006" />. | ||
| + | |||
| + | In general, lower storage temperatures preserve hop compounds. Cans also help preserve some hop compounds versus bottles because bottle caps can strip certain hop compounds such as myrcene and caryophyllene when stored at room temperature (less so when stored cold). For example, one study found a moderate amount of degradation of humulinones, iso-α-acids, and residual α-acids when dry hopped beers were stored at 20°C versus 3°C. There was also an overall decrease in hop aroma compounds during warm storage, with some esters, hop monoterpenes, and sesquiterpenes showing poor storage stability compared to other ester compounds, monoterpene alcohols, and ketones which increased during warm storage. After 10 months of storage, the dry hopped beers stored at 20°C had a significant drop in floral, citrus and tropical fruit notes when compared to the same beers stored at 3°C <ref>[https://onlinelibrary.wiley.com/doi/full/10.1002/jib.667 Kemp, O., Hofmann, S., Braumann, I., Jensen, S., Fenton, A., and Oladokun, O. (2021) Changes in key hop-derived compounds and their impact on perceived dry-hop flavour in beers after storage at cold and ambient temperature. J. Inst. Brew., https://doi.org/10.1002/jib.667.]</ref>. | ||
[[Tetrahydropyridine]] (THP) is a compound that tastes like Cheerios® or corn tortilla chips that often develops soon after packaging beers that contain ''Brettanomyces'' or heterofermentative ''Lactobacillus''. It is usually detected after swallowing the beer. This compound is stimulated by oxygen, and often ages out after a few months. See the [[Tetrahydropyridine]] page for more information. | [[Tetrahydropyridine]] (THP) is a compound that tastes like Cheerios® or corn tortilla chips that often develops soon after packaging beers that contain ''Brettanomyces'' or heterofermentative ''Lactobacillus''. It is usually detected after swallowing the beer. This compound is stimulated by oxygen, and often ages out after a few months. See the [[Tetrahydropyridine]] page for more information. | ||
Revision as of 01:16, 24 December 2023
Overview of goals: https://www.facebook.com/groups/MilkTheFunk/permalink/1640076039353937/
BEGIN ROUGH DRAFT
(In progress) For this page, Aging and Storage will refer to the conditioning and aging of beer in its final package (bottle, keg, etc.). The conditioning process includes the changes that take the beer from its state at packing to the state in which it is intended to consumed. The term aging will be used on this page to discuss changes in the conditioned beer as it is aged further. Storage conditions and their advantages and disadvantages will be discussed. Aging of beer before packaging is discussed in various brewing pages on the wiki and will not be discussed here.
For simplicity, this page will mostly refer to what is going on in a bottle, but the same changes and processes occur in other package types, albeit at different rates, and 'bottle' can be replaced with 'keg' or another final package.
Contents
- 1 Best Practices for Storage
- 2 Bottle conditioning
- 3 Techniques of Cellaring
- 4 Microbial Survival and Changes
- 5 Sensory Changes
- 6 Chemical Changes
- 7 See Also
- 8 References
Best Practices for Storage
This could be an overview for customers, retailers, and distributors. The sections below can give more technical/detailed information.
See Techniques of Cellaring below for more information.
Bottle conditioning
(in progress)
Bottle conditioning is the process and changes that take a beer at packaging time to beer that is ready to drink. This can include the development of carbonation, microbial growth, development and reprocessing of off flavors, 'bottle shock' and other changes. Bottle conditioning, at least for the initial period where carbonation is generated, is typically carried out at warmer temperatures than extended aging after the conditioning is done.
See also:
- "Bottle Conditioning," Topic Review by Kateřina Štulíková and Pavel Dostálek, Scholarly Community Encyclopedia.
- Packaging and Oxygen Exposure.
- Justin Amaral's can conditioning project with DO tracking for canned conditioned beers on MTF.
Techniques of Cellaring
Cellaring, or extended age in the bottle once the beer is ready to drink, is common for many mixed fermentation beers. Cellaring is typically carried out at cooler temperatures.
Bottles vs Kegs vs Cans vs PET
Chemical changes over time can be different as the beer ages. The packaging type can have a significant impact on how the beer ages. One study on lager found that PET bottles had the greatest variation in chemical changes over a 6 month period of time at ~20°C compared to glass bottles, kegs, and cans. In particular, diacetyl was higher in PET bottles as the beer aged. This was attributed to the PET bottles being more permeable to oxygen because of oxidation of acetoin and 2,3-butanediol to form diacetyl, or the oxidative decarboxylation of alpha-acetolactate, a precursor to diacetyl. Cans showed the least formation of diacetyl, while kegs and bottles displayed moderate increases in diacetyl over time compared to the PET bottles. Acetaldehyde was also highest in the PET bottles. DMS had a high spike during the first month of storage, but by the end of 6 months, the PET bottles had less DMS than the other storage types and glass bottles had the most. Other compounds had less significant differences between package type (ethyl acetate, propanol, isobutanol, isoamyl alcohol, higher alcohols, and esters) [1].
Corks vs Caps
There haven't been many studies on the aging of beer packaged in corked bottles. There have been quite a few studies on Champagne and sparkling wine that have looked at various aspects of aging in corked bottles, and these studies might help us understand some aspects of beer stored in bottles that are corked.
CO2 Loss Over Time
Young finished champagne and sparkling wines produced according to the méthode traditionnelle process, which involves carbonating the champagne with sugar for 15 months and then disgorging them and corking them, begin with a CO2 concentration of around 11-12 g/L (~6 volumes), while sparkling wines that are 5 years old and 10 years old have been found to have a much lower concentration of CO2 at around 6-8 g/L (~3-4 volumes) [2][3]. The gradual loss of carbonation in sparkling wines has been attributed to the porous nature of corks allowing for the slow diffusion of gasses through them, which is highly variable based on the density of the cork [4][5], as well as the interface between the cork and the neck of the bottle [3]. An interesting observation is that there wasn't a large difference in carbonation loss between 5-year-old sparkling wines and 10-year-old sparkling wines, indicating that the loss of carbonation could greatly slow down once the liquid inside reaches around 3-4 volumes of CO2.
The construction of the cork itself is a variable that makes it difficult to predict the exact rate of CO2 loss. Corks are composed of two distinct parts: the mushroom of the cork is made up of agglomerated cork small granules, while the foot of the cork is made up of two large cork slices. This lower part is made up of several lenticels, which are parts of the plant that allow gasses to flow in and out of the plant. These lenticels vary from cork to cork. Nevertheless, a model has been proposed by Liger-Belair et al. that estimates the amount of CO2 loss over time. In this model, two other variables have been identified as playing a large role in how much CO2 is lost: storage temperature and bottle size. The warmer the storage temperature, the faster the rate is of losing of CO2, and the larger the bottle volume the slower the rate is of losing CO2. Below are some estimated CO2 levels based on the Liger-Belair model in g/L and then converted to volumes in parenthesis at various points in time. The first table shows the estimated amount of CO2 loss when stored at three different temperatures (4 °C, 12 °C, and 20 °C). The second table shows the estimated amount of CO2 loss in different sized bottles (1.5 L, 750 mL, and 350 mL) when stored at 12 °C [3]:
| Storage Temp °C [3] | CO2 g/L (vols) at Year 0 | CO2 g/L (vols) at Year 5 | CO2 g/L (vols) at Year 10 | CO2 g/L (vols) at Year 15 | CO2 g/L (vols) at Year 20 |
|---|---|---|---|---|---|
| 4 | 12 (6) | 9.5 (4.75) | 7.5 (3.75) | 6 (3) | 5 (2.5) |
| 12 | 12 (6) | 9 (4.5) | 6.5 (3.25) | 5 (2.5) | 4 (2) |
| 20 | 12 (6) | 8 (4) | 5.5 (2.75) | 4 (2) | 2.5 (1.25) |
| Bottle Size stored at 12 °C [3] | CO2 g/L (vols) at Year 0 | CO2 g/L (vols) at Year 5 | CO2 g/L (vols) at Year 10 | CO2 g/L (vols) at Year 15 | CO2 g/L (vols) at Year 20 |
|---|---|---|---|---|---|
| 1.5 L (Magnum) | 12 (6) | 10 (5) | 8.5 (4.25) | 7 (3.5) | 6 (3) |
| 750 mL (Standard) | 12 (6) | 9 (4.5) | 6.5 (3.25) | 5 (2.5) | 4 (2) |
| 375 mL (Half Bottle) | 12 (6) | 7.5 (3.75) | 5 (2.5) | 3.5 (1.75) | 2 (1) |
The above tables show a significant decrease over the first 5 years, and then a gradual decrease over the decades. As the pressure decreases inside the bottle, the rate of CO2 loss slows down. This data may or may not accurately reflect highly carbonated sour beer. Other factors in sour beer that do not apply to sparkling wine and Champagne include living Brettanomyces which could continue to slowly release CO2 by fermenting long chain sugars, although this could also be an insignificant amount if the beer was aged for a long time before packaging (see Final Gravity). Similar studies in beer have not been published as far as we know.
Bottle Orientation
A study on still white wines found that in some cases, bottles stored on their sides had slightly less oxidation after 5 years or storage than if they were stored upright, however, the larger variable in the study was the type of cork (natural vs synthetic, with the synthetic cork allowing more oxidation over 5 years of storage than the natural corks). It has long been asserted that still wine should be stored on its side so that the cork remains wet, which expands the cork and limits oxidation over time. However, this study found that cork wetness and bottle orientation had only a small impact in how well the wine aged, and the two types of corks studied differed in how wet they were when stored upright. When comparing two different types of corks for the same wine and stored upright versus on their side, one cork type had relatively low wetness regardless of bottle orientation (~15% after 5 years), while the other cork type was less wet when stored upright (65% upright vs 90% horizontal after 5 years). Regardless of the wetness of the corks, they performed similarly as far as protecting the wine from oxidation regardless of the wetness and bottle orientation (bottle orientation had minor effects in some cases), however, a previous study found that wine bottles stored on their sides were preserved better [6]. See also this MTF discussion. Inferring any results from these studies to carbonated beer is difficult because the internal pressure caused by carbonation will create a different dynamic of gas exchanges through the cork (see Corks vs Caps above). However, this information might be helpful in understanding the wider picture, and it is directly relevant to uncarbonated beer that is stored in corked and caged bottles, such as some Belgian lambics or uncarbonated American sour beers.
In beer brewing, horizontal storage might only be done during the initial conditioning phase after first being bottled (1-3 months), and then additional storage would be be vertical. Vertical storage will allow yeast and other compounds to settle to the bottom of the bottle and might be more ideal for long term storage than horizontal storage [7] (~31:00).
- Brandon Jones article on corked bottle storage (verticle vs horizontal) with comments from Belgian lambic brewers.
- Pierre Tilquin explains that horizontal storage for corked bottles results in no Pellicle formation in the bottle [8]. Also reported in a side by side comparison by James Howat.
- The internal pressure caused by CO2 inside the bottle keeps the cork moist without needing to lay the bottle on its side. Uncarbonated lambic-style beers might benefit from horizontal storage by keeping the beer in contact with the cork and keeping it moist (this doesn't matter for synthetic corks because they don't absorb moisture) [9][10][11]. However, other sources claim that the humidity in the headpace (ullage) of uncarbonated wine is close to 100% even when stored upright [12][13][14].
- Jeff Porn observes accelerated flavor development (good or bad) when comparing bottles that are stored horizontally versus ones stored vertically. Interestingly, bottles stored on their side developed Tetrahydropyridine off-flavor while the vertically stored bottles did not (Jeff bottle conditions with a house culture) [15]. Shawn Savuto also reported THP development only in bottles aged on their side [16]. Increased flavor development was also reported by Brasserie Dupont.
- Blake Tyers from Creature Comforts reported no statistical significance been horizontal and vertical storage, however those that did identify a difference correctly noticed harsher flavors and "more edges" in the bottles stored upright.
- Ryan Fields reported not seeing a difference between horizontal versus vertical storage, however, they only tested this one time. He still prefers to age bottles vertically in cages because this requires less space than aging in stacked boxes, and aging in boxes can insulate from proper airflow and temperature [7] (~29:45).
- Alex Levy reported off-flavors in MTF when storing bottles that were capped (no cork) horizontally but not vertically.
- For equipment and methodology of using wire cages to store horizontally, see Wire Storage Containers.
Microbial Survival and Changes
Cover what we know the about survival rate of different microbes, and connect them to the above sections if they have an impact.
Brettanomyces is regarded as an "oxygen scavenger", and can survive the harsh conditions of packged beer for many years. It is believed that any micro-oxidation that occurs in bottles will be consumed by any living Brettanomyces. The oxygen scavenging capabilites of Brettanomyces might be impacted by the temperature at which the bottles are stored. For example, colder conditions might prevent them from consuming oxygen, although more data is needed to show that this is true.
MTF thread with Richard Preiss about isolating microbes from old Berliner
Spitaels et al., 2015 microbes in bottles of gueuze
Anecdotal evidence that Brettanomyces can reverse discoloration from enzymatic browning.
See also Commercial Sour Beer Dregs Inoculation.
Sensory Changes
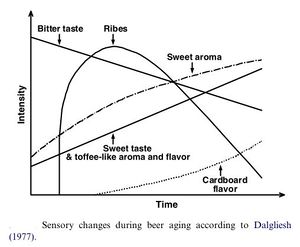
Beer aging is a complicated subject, and every beer ages differently. In general, sensory changes include a slow and gradual and steady decline in bitterness, partly due to an increase in a honey/toffee-like sweetness in the aroma and taste. Another flavor described as 'ribes', which is a sort of black currant taste, rises quickly sometime after packaging, and then gradually declines over time. After the 'ribes' flavor, a cardboard flavor slowly develops. Other findings claim that the cardboard flavor increases to a maximum, and then begins to decline after some time. Other changes include a rise in harsh, astringent notes in some beers, while wine and whiskey notes develop in very old beers. Positive flavors such as fruity esters and hop character fade considerably. These changes are not always seen as negative changes; studies have been done that show that drinkers sometimes prefer aged beers over young ones (even in light lagers). While the cardboard flavor has historically been emphasized in beer staling, it is just one component of beer aging. Many other flavor changes can occur, depending on the beer. For example, stronger beers have been found to develop burn, caramel, alcohol, licorice, and Sherry characters and no cardboard flavors. The development of many of these flavors corresponds with the amount of dissolved oxygen in the packaged beer and storage temperature [18].
Chemical Changes
(In Progress)
As beer ages, different flavor and aroma compounds both deteriorate and form over time and at different rates. This is a complex collection of changes with many variables involved in affecting different compounds in different ways. As molecules deteriorate or are formed, their associated flavors not only dissipate and appear, but the influence that they have on each other may emphasize or overwhelm other flavors. The compounds, which number in the hundreds or perhaps thousands, are not in equilibrium when the beer is packaged. Once bottled or kegged, this closed environment forces molecules to reach a status of minimal energy and maximal entropy, and thus many of the molecules change over time to reach equilibrium. The two largest factors that affect beer aging are temperature and dissolved oxygen in the package [18]. Regular beers with a lower pH tend to age faster, however, most beer studies have focused on lager beers and some on ales and strong beers. Research on beers containing living Brettanomyces or beers that are at a lower pH of 3-3.8 is limited.
Overview of compounds formed in beer during storage [18]:
- Linear aldehydes: acetaldehyde, trans-2-octenal, trans-2-nonenal, derivatives of nonadienal and decadienal.
- Strecker aldehydes: 2 and 3-methyl-butanal, 2-phenylacetaldehyde, benzaldehyde, 3-(methylthio)propionaldehyde (also known as methional).
- Ketones: trans-beta-damascenone, 3-methyl-2-butanone, 4-methyl-2-butanone, 4-methyl-2-pentanone, diacetyl, 2,3-pentanedione.
- Cyclic acetals: 2,4,5-trimethyl-1,3-dioxolane, 2-isopropyl-4,5-dimethyl-1,3-dioxolane, 2-isobutyl-4,5-dimethyl-1,3-dioxolane, 2-sec butyl-4,5-dimethyl-1,3-dioxolane.
- Heterocyclic compounds: furfural, 5-hydroxymethyl-furfural, 5-methyl-furfural, 2-acetyl-furan, 2-acetyl-5-methyl-furan, 2-propionylfuran, furan, furfuryl alcohol, furfuryl ethyl ether, 2-ethoxymethyl-5-furfural, 2-ethoxy-2,5-dihydrofuran, maltol, dihydro-5,5-dimethyl-2(3H)-furanon, 5,5-dimethyl-2(5H)-furanon, 2-Acetylpyrazine, 2-methoxypyrazine/2,6-dimethylpyrazine, trimethylpyrazine, tetramethylpyrazine.
- Ethyl esters: ethyl 3-methyl-butyrate, ethyl 2-methyl-butyrate, ethyl 2-methyl-propionate, ethyl nicotinate, diethyl succinate, ethyl lactate, ethyl phenylacetate, ethyl formate, ethyl cinnamate.
- Lactones: nonalactone, hexalactone.
- Sulfur compounds: dimethyl trisulfide (production enhanced by low pH), 3-Methyl-3-mercaptobutylformate.
General Effects of Oxidation
Oxidation, also known as a redox reaction, is the chemical process of atoms losing electrons to other atoms. The atom that loses an electron is called the "oxidizer", and the atom that gains the electron is called the "reducer". Despite the name of this process being called "oxidation" and oxygen often being the reducer, oxygen is not required since other chemicals can serve this purpose. Oxidation can occur slowly (e.g. metal rusting) or quickly (e.g. fire), and applies to a large range of simple and complex processes [19]. In beer, oxidation takes the form of carbon-based molecules or metal ions losing electrons to either oxygen molecules or free radicals. Oxygen itself in its ground state is not particularly reactive in beer, however, oxygen in beer reacts with transition metal ions found in beer such as copper, iron, and manganese (these include two types of redox reactions called the Fenton, and Haber-Weiss reactions) to form "reactive oxygen species" (ROS) which then react with other compounds in the beer to cause staling [20]. Oxidation reactions increase the amount of off-flavor compounds, as well as dulls the aroma of beer. Examples of off-flavors produced by these redox processes include aldehydes and ketones (e.g. acetaldehyde), trans-2-nonenol, and diacetyl. Beer also darkens in color when exposed to oxygen, probably through an oxidation process known as enzymatic browning. Brewers yeast and Brettanomyces are great scavengers of oxygen, and adding fresh yeast and sugar at packaging can help reduce dissolved oxygen in the package, and even reverse some effects of oxidation [21][22][20].
In general, the best practice is to limit dissolved oxygen (DO) levels to 30-60 ppb (or 40-150 ppb total package oxygen (TPO)) [23], much of which is picked up at packaging time, although brewers have had success packaging beers with living Brettanomyces without purging the bottles with CO2. Dissolved oxygen should be measured with a dissolved oxygen sensor/meter during production and immediately at packaging; Bradley Barnette's Masters thesis showed that beers that were oxygenated at packaging to test the effect on dry hopping at 200 ppb had similar DO levels to beers oxygenated at 50 ppb after two weeks of storage, which lead to the hypothesis that oxygen is consumed via oxidation reactions over time during the storage of beer [20]. Other compounds can serve as anti-oxidants in beer. For example, sulfates are converted into sulfites by yeast, and sulfites postpone the formation of free radicals. Lower-weight polyphenols, which originate from malt (70-80%) and hops (20-30%), are thought to be free radical scavengers and anti-oxidants, however other polyphenols have been identified as pro-oxidants and the effectiveness of antioxidant activity in general for polyphenols is debatable in the scientific literature (although their impact in the mash and boil has been established as positive). Maillard reactions from malting/roasting and wort boiling also create anti-oxidants, and in general the darker the roasting the more anti-oxidant the malts will be [18], although compounds in kilned malts, hypothesized to be the proanthocyanidins and flavonols derived from Maillard reactions, have been found to be a source for oxidation and beer staling. Alpha acids and iso-alpha acids have been shown to react with transition metal ions (iron), thus reducing the impact of the oxidation of iron ions [20]. Lactic acid and lactic acid fermentation are thought to also help serve as anti-oxidants, although this has not be studied in sour beer [24][25].
Oxygen has a large and negative impact on highly hopped beers. Dry hopping also serves as a greater risk of oxidation in beer. Oxygen can enter the beer during the dry hopping process. It's also been shown that iron ions increase from hop additions, which react with oxygen and cause oxidation. Higher DO can slightly increase the rate of IBU degradation. Interestingly, during two weeks of storage it was shown by Barnette that hop compounds such as monoterpenes did not decline even though flavor analysis reported decreased hoppy, fruity, and citrus character, suggesting that this is caused by the production of other oxidized compounds in the beer rather than a decrease hop-derived compounds [20].
See also:
- "Controlling Beer Oxidation" by George Fix.
- Industry standards of dissolved oxygen levels in beer throughout the brewing process, by Hach.
- MTF thread on anecdotal accounts of Brettanomyces affecting oxidation character in beer.
- Dr. Bryan Heit explanations chemical oxidation pathways, and the use of metabisulfite to limit oxidation in packaging.
General Effects of Temperature
The temperature at which beer is stored has also has a major impact on how beer ages. The effect that temperature has on a given reaction depends on the type of reaction; not all reactions are increased at the same rate. For example, it has been reported that beer stored at 25°C tends to develop more caramel flavor, while the same beer stored at 30°C develops more cardboard flavor [18]. Bamforth and Lentini proposed a simplified version of the Arrhenius model known as Q10 to generalize the rate of chemical reactions in beer based on temperature. Q10 is a measure of the temperature sensitivity of chemical and enzymatic reactions due to an increase in temperature by 10°C. The equation is expressed as a ratio: Q10 = Reaction Time + 10°C / Reaction Time. Bamforth recommends that for beer, Q10 will be 2 or 3 (most chemical reactions fall in this range). For example, assuming a Q10 of 3, storing beer at 30°C for 2 weeks is equivalent to 6 weeks at 20°C, 18 weeks at 10°C, and 54 weeks at 1°C. Assuming a Q10 of 2, storing beer at 30°C for 2 weeks is equivalent to 4 weeks at 20°C, 8 weeks at 10°C, and 16 weeks at 1°C [26][20][27]. Barnette's Masters thesis found that warmer temperatures had a greater negative impact on hop flavor and aroma than high levels of dissolved oxygen over a two week storage period in dry hopped beers [20].
Haze
Also referred to as colloidal instability, haze has been deemed a problem for most beer styles for mainly aesthetic reasons, especially for lighter styles of beer where haze is more noticeable than darker styles. While clarity is considered important for most beer styles, some beer styles are intended to be hazy (some German wheat beers, Belgian wheat beers, NEIPA, Abbey ales that are refermented in the bottle, some Belgian lambic and derived beers, and some mixed fermentation beers). Clarity in packaged beer is one form of beer stability (along with flavor/aroma, color, and foam stability). Haze can be caused by wild yeast and lactic acid bacteria, carbohydrates, binding of proteins and oxidized polyphenols, metals such as iron, oxidation, aldehydes, shaking, exposure to high temperatures, exposure to polyphenol-rich materials, and sunlight [28][29].
Polyphenol-protein haze is the most common cause of haze formation after packaging. It comes in two forms: chill haze and permanent haze. Chill haze (or reversible haze) is the combination of polyphenols and proteins via non-covalent bonds at colder temperatures. This haze generally goes away after the beer is warmed up again. When beer remains hazy at room temperature (specifically 20°C), chill haze becomes permanent haze [28][29].
Beer contains much more haze-active proteins than haze-active polyphenols. Haze-active proteins are acidic hydrophilic polypeptides that originate from barley and are rich in proline, glutamic acid, and glycosylated. Smaller phenols such as phenolic acids and flavonols do not contribute to haze, but heavier oxidized polyphenols such as procyanidin and prodelphinidin are strong permanent haze inducers. These proteins and oxidized polyphenols bind together to form haze in beer [28]. The polyphenol found in hops, catechin, does not form haze immediately but can cause haze after a period of storage. The pH of the beer has a huge impact on this reaction: much more haze is formed at a pH of 4.0 than it is at a pH of 3.0 or above 4.2. Higher ABV beers also encourage more haze formation from proteins and polyphenol reactions to cause permanent haze. Permanent haze from proteins and oxidized polyphenols generally form after a period of storage (called the "lag phase"); the longer this lag phase, the better the beer's colloidal stability [29].
Brewers generally remove haze by additives such as tannic acid, papain, or silica gel. Some of these additives can also remove foam forming proteins. Because of this, a chemical known as PVPP is often used because it does not remove foam forming proteins. Several products are available that contain combinations of PVPP and other compounds [29].
Acids and Esters
Cover microbiologically driven changes: over-attenuation, Brett expression under pressure, autolysis
pH change in the bottle?
http://www.sciencedirect.com/science/article/pii/S0740002014002548
Some volatile esters that give the beer a fruity flavor, such as isoamyl acetate that gives the beer a banana flavor, can decrease to levels below their flavor thresholds over time in packaged beer. Other volatile esters such as ethyl 3-methyl-butyrate (derived from oxidized alpha and beta acids in hops and contributes a wine-like character [30]), ethyl 2-methyl-butyrate (derived from oxidized alpha and beta acids in hops and contributes a wine-like character [30]), ethyl 2-methyl-propionate, ethyl nicotinate, diethyl succinate, ethyl lactate, ethyl phenylacetate, ethyl formate, ethyl furoate and ethyl cinnamate (fruity, sweet character) are formed during beer aging. Lactones, which are cyclic esters, such as the peach-like hexalactone and nonalactone tend to increase during beer storage and have a significant impact on the flavor of aged beer [18]. See also Esters From Hops.
While most ester formation and hydrolysis (breakdown) in beer during aging are mostly acid-catalysed, some of this activity is due to esterase enzymes. It has been shown that some esterases are released by yeast autolysis during beer aging. This release of esterase enzymes is strain dependent, with ale yeast having more potential for this activity than lager yeast. The enzyme is most active at 15-20°C and is destroyed by pasteurization [18].
Phenols
Phenols are a large class of organic compounds. One way that phenols can be classified is by how many carbon atoms they include (see the phenols Wikipedia article). Examples of classes of phenols include the phenol (the simplest form of phenols with 6 carbon atoms), hydroxycinnamic acids (ferulic acid, caffeic acid, etc.), and complex polyphenols (multiple phenol structure units) [31][32].
Many phenols have an impact on beer aging or are impacted by beer aging. They are introduced from malt, hops, and yeast fermentation, as well as from some brewing adjuncts or techniques (additions of fruit, barrel aging, etc.). Some phenols directly impact the flavor, astringency, haze, body, and fullness of beer. Some phenols also have health properties. Degradation of some phenols leads to the changing of fresh beer taste. Other phenols act as antioxidants and can protect the beer to some degree from oxidative degradation as beer ages [29].
Phenolic Monomers
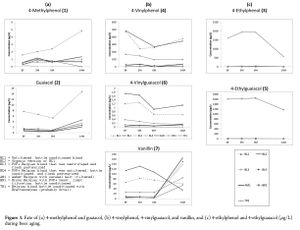
Phenolic monomers include phenolic acids (also known as "hydroxycinnamic acids"), flavonols, and volatile phenols (4-vinylphenol, 4-vinylguaiacol, 4-vinylcatechol, and their ethyl derivatives) [34].
Hydroxybenzoic acids (e.g. vanillic acid, gallic acid, syringic acid) and hydroxycinnamic acids (e.g. p-coumaric, acid, ferulic acid, sinapic acid, caffeic acid) are extracted from polysaccharides within the cell walls of malted grains during the mashing process, and are generally considered to have some antioxidant qualities, however at least one study found that they did not positively impact the oxidative reactions in aging beer (some phenolic acids are antioxidants, but others are oxidizers, and the net result is possibly a non-effect of oxidation and preventing oxidation). They generally do not impact flavor because of their high flavor threshold in beer (52 ppm for p-coumaric acid, 66 ppm for ferulic acid - these acids are generally bitter and astringent in flavor when above flavor threshold), however yeast metabolism can lead to flavorful volatile phenols such as 4-vinylphenol (plastic) and 4-vinylguaiacol (clove flavor; 0.3 ppm flavor threshold in beer) [34][29]. 4-vinylguaiacol can be partially oxidized or reduced into smaller compounds such as vanillan, 4-ethylguaiacol (reduced by Brettanomyces), and guaiacol as beer ages [29].
Volatile phenols include 4-vinylphenol, 4-vinylguaiacol, 4-vinylcatechol, and their ethyl derivatives which are produced by Brettanomyces. Although the production of volatile phenols during fermentation has been heavily researched, the aging of volatile phenols in bottled beer hasn't received much attention from science. One study looked at the evolution of various volatile phenols in several Belgian beers, including one Trappist beer that was conditioned with Brettanomyces (probably Orval). All beers were aged at 20°C/68°F in a dark room. The study found that Belgian beers that did not contain Brettanomyces and that had a high level of 4-vinylguaiacol (4VG) saw a drop in 4VG of about 50% between months 3 and 6, and then a very slow increase from months 6 to 14. This also corresponded with a fairly sharp increase in vanillan beginning after 6 months. This decrease in 4VG and increase in vanillan was suggested to be caused by both the oxidation of 4VG and acid hydrolysis of glycosides. Both guaiacol (roasted coffee flavor) and 4-methylphenol (burnt flavor) saw a sharp rise after 6 months to 14 months of aging, particularly in the dark Belgian beers (from 6 to 15 ug/L and 2 to 5 ug/L respectfully). The phenols guaiacol and 4-methylphenol have been associated with darker roasted malts [33]. The degradation of 4VG has also been reported to be 25% after 20 days at room temperature, or 50% after 20 days at 40°C/104°F [29].
For the Belgian beer that was conditioned with Brettanomyces, 4-ethylphenol (4EP), which is responsible for the plastic off-flavor in beers and wine with Brettanomyces, steadily increased from 1500 ug/L to 2000 ug/L from when bottled to 3 months. From month 3 to 6, the level of 4EP was stable. From month 6 to 14, 4EP showed a steady decline from 2000 ug/L to 500 ug/L, indicating that 4EP can age out of bottled beers. The phenol 4-ethylguaiacol (4EG) was steady at 1600 ug/L with only a very slight increase from bottling date until 6 months. From month 6 to month 14, 4EG dropped from 1600 ug/L to 1200 ug/L. This indicates that 4EG is relatively stable compared to 4EP, but is not immune to breaking down [33].
The volatile phenol 4-vinylsyringol (smokey, burnt) has been identified in lagers that have aged and is thought to stem from the slow acidic hydrolysis of a glycoside during aging [29].
Polyphenols
Polyphenols are a large group of organic chemicals characterized by many phenol structures combined. Subclasses of polyphenols include tannic acid, tannins, and flavonoids [32][35].
Polyphenols have an ambiguous role in the aging of beer. Flavonoids (for example catechin, which comes from hops and is a major source of polyphenols in beer), are antioxidants and protect more sensitive compounds such as isohumulones from oxidation by scavenging free radicals and binding with oxidative metals (iron, for example). However, they themselves can also be oxidized over time to possibly create off-flavors. In addition to their own oxidation, hydroxyl radicals that cause oxidation also react highly with ethanol, and therefore a portion may not react with polyphenols. After a lag period of 5 weeks in the bottle, it was found that levels of tannins actually increase. This is thought to be caused by smaller flavonoids reacting with acetaldehyde. Polyphenols were also oxidized into quinones, which are a stepping stone in the reaction that causes oxidative food browning (this reaction increases in the presence of acids [36]). The use of polyphenols during mashing and boiling has been shown to decrease trans-2-nonenal (cardboard flavor) and trans-2-nonenal that is protected from fermentation by being bound to proteins (see Tannic Acid below). In two studies, there appeared to be no significant effect on free radical formation by polyphenols, probably due to the fact that they readily react with ethanol [29].
Higher temperatures increase the rate of oxidized polyphenols. In one study on aged lagers, 6.5% of the polyphenols were oxidized after 5 days at 40°C/104°F, but only 0.6% of the polyphenols were oxidized after 9 months at 20°C/68°F [29].
Polyphenols generally contribute to an astringent taste in beer, and this can be intensified at a lower pH (4-4.2). Oxidation of polyphenols might make them more astringent depending on the degree of "polymerization degree" (see this article), although residual sugars reduce their astringency. Sensory analysis of lagers has shown that aged lagers became less bitter and more astringent over time (especially at a higher temperature or a higher pH), probably due to a decrease in IBU's and bitter polyphenols like catechin, and an increase in oxidized polyphenols [29].
Heavier polyphenols such as procyanidin and prodelphinidin also contribute to beer haze (see Haze above) and darkening in color after packaging. While beer darkening is also caused by oxygen and storage at higher temperatures, and possibly also by slow Malliard reactions, yellow-brown pigments can also come from oxidized polyphenols. The catechin polyphenol from hops can oxidize into yellow pigments such as dehydrodicatechin. This oxidative reaction occurs more so at a pH of 6 rather than at a pH of 3, and is greatly inhibited by storing beer cold. These reactions have been studied more so in wine and port; anthocyanidins bind with flavanols or acetoacetic acid, producing yellow-brown pigments. Pyruvic acid, vinylphenol, cinnamic acid, glyoxylic acid, ad furfural derivatives have also been identified as precursors to yellow-brown pigmentation in wine [29].
Tannic Acid
Tannic acid is a subclass of tannin (tannins are a subclass of polyphenols). Tannic acid is generally extracted from four sources: Chinese gallnuts, Aleppo gallnuts (these both produce gallic acid), Sumac leaves, and Tara pods (produces quinic acid). Forms of tannic acid can also be extracted from oak galls (seeds from oak trees) and oak bark [37]. Tannic acid extracts are of interest to the brewing industry because they have been found to improve flavor stability through its antioxidant Properties. Specifically, these properties include free radical scavenging activity, bonding of the metals involved in beer staling (specifically iron ions and perhaps copper ions), and preventing oxidative degradation of lipids that produce the aldehydes such as trans-2-nonenal (trans-2-nonenal is responsible for the paper/cardboard off-flavor in stale beer). Tannic acid also increases colloidal stability (clarity over time) by binding with the proline-rich proteins that cause chill haze. The higher molecular weight tannic acids (Chinese gallnuts or Sumac leaves) will reduce chill haze by removing these proteins, while medium molecular weight tannic acids (Allepo gallnuts) can result in a stable haze for beer styles where some haze is desirable such as Belgian Wit or German Hefeweizen [38].
One study showed that adding gallic acid (tannic acid from Chinese or Aleppo gallnuts) during the mash and sparge water extended the shelf life of beer by reducing off-flavors such as trans-2-nonanol, the aldehyde responsible for the papery taste in stale beer. It also bound to proteins containing thiols that when oxidized during mashing can cause filtering and amylolytic issues, and settled them out during the mashing/boiling which made filtration easier. The study determined that the tannic acid did not have an impact on the saccharification rest or the soluble protein content (including head retention proteins). It did have a very slightly negative impact on yeast attenuation, however the addition of zinc resolved the attenuation issue (it is thought that the gallic acid dropped the zinc out of solution since it binds with metals, and thus negatively impacted the yeast's health) [39].
The sensory analysis in this study showed that all of the fresh beers with or without gallic acid scored positively by tasters, although they preferred the beers with gallic acid because of their "fullness of taste and mouthfeel". After 5 days of storage at 40°C, the beers with gallic acid continued to rate high while the non-gallic acid beers began to show signs of staling (darker in color, cardboard flavor, slight sweetness, and a sharp unpleasant bitterness). After storing the beers for 10 days at 40°C the non-gallic beers were clearly more oxidized. These results were confirmed by also storing the beers for one year at 4°C in the dark; the beers with gallic acid were always preferred by tasters. The beers with gallic acid added during the mashing and sparging had better flavor stability and better stability of hop compounds such as iso-alpha acids. They also had a higher polyphenol content from the malts (only hop extract was used, so the polyphenol content had to come from the malts). The lipoxygenase activity (LOX), which is the mechanism by which lipids are oxidized to create off-flavors, was also decreased in the beers with gallic acid added. They also found that the "Strecker" and "Maillard" derived aldehydes phenylacetaldehyde, furfural, benzaldehyde were reduced by the use of gallic acid (fatty-acid derived aldehydes were not reduced, but there is evidence that these are already present in malt and are not formed during the mashing process, unlike the previously mentioned aldehydes). Trans-2-nonenal (paper, cardboard staling flavors) was also reduced in beers brewed with gallic acid. The study found that mashing with gallic acid at 62°C and 5.3 pH had the most positive impacts on both flavor stability and the prevention of off-flavor development [39].
Tannic acid products are now being sold in the brewing industry, such as Brewtan B which contains gallic acid extracted from oak galls without the impurities that can often be found in other commercial tannic acid products. This manufacturer claims that the product enhances initial clarity of wort and colloidal stability of beer over time, stabilizes hop bitterness, reduces aldehyde formation, and increases flavor and color stability over time. The dosage for this product is quite low at 1.5–4.0 g/hL [38]. Author Denny Conn of Experimental Brewing has noted that the use of Brewtan B increases the mouthfeel and flavor stability of beer [40]. Brewtan B specs and usage can be found here.
See also:
Hop Compounds
- https://www.tandfonline.com/doi/full/10.1080/03610470.2019.1705037?needAccess=true - https://www.tandfonline.com/doi/abs/10.1080/03610470.2020.1843898
IBU Degradation
Iso-α-acids, which are the main component of bitterness in beer, degrade over time in the bottle. Iso-α-acids are comprised of trans isomers and cis isomers of isohumulone, isocohumulone, and isoadhumulone. The trans forms of these iso-α-acids degrades much faster than the cis versions. This degradation is attributed to oxidative reactions. According to one study this included oxidative reactions in the mash when the mash was exposed to oxygen versus flushed with CO2. Although the difference was slight, the mash that was exposed to oxygen had slightly more iso-α-acids degradation over time in the packaged beer than a mash that was flushed with CO2 (this degradation was emphasized more with time rather than hotter temperature once packaged). Beers stored at 20°C had their trans iso-α-acids degrade gradually (10% at day 20, 25% at day 50, 35% at day 100, and 45% at day 200), while cis iso-α-acids stayed stable. This rate of decay was nearly tripled when the beer was stored at 30°C. The ratio of remaining trans iso-α-acids to the more stable cis iso-α-acids also corresponded to the stale flavor of beer, and thus this ratio was offered as a quantitative measurement of the oxidation/staling of beer [41]. The cis isomers are generally considered more bitter than the trans isomers, so aged beer will still maintain some amount of bitter character [42].
Lightstruck
- https://beersensoryscience.wordpress.com/2011/03/17/lightstruck/
- http://www.scielo.br/scielo.php?pid=S0100-40422000000100019&script=sci_arttext&tlng=es
- http://onlinelibrary.wiley.com/doi/10.1002/j.2050-0416.2002.tb00568.x/abstract
- http://www.professorbeer.com/articles/skunked_beer.html
- https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/1521-3765%2820011105%297%3A21%3C4553%3A%3AAID-CHEM4553%3E3.0.CO%3B2-0
- https://pubs.rsc.org/en/content/articlelanding/2004/pp/b316210a/unauth#!divAbstract
Iso-alpha acids will skunk if exposed to UV light. Oxidized alpha acids (humulinones) will also skunk if exposed to UV light [43]. This compound is known as 3-methyl-2-butene-1-thiol (3MBT). Brown bottles filter most UV light, while green bottles only filter a portion of UV light. See "Lightstruck", Craft beer and Brewing Magazine website.
Damascenone
The ketone beta-damascenone (rhubarb, red fruits, stewed apples; threshold of 25 ppb [22]) is thought to be formed from the oxidation of hop oils, although it has also been found in unhopped aged beers. Potential precursors are allene triols and acetylene diols formed from the degradation of the carotenoid neoxanthin. It was found to increase greatly in aged beer that was at a pH of 3 or 4.2 versus a higher pH. This was attributed to the acidic hydrolysis of glycosides. The release of flavor compounds from glycosides could be present in acidic beers that are aged on fruit or herbs [44][18]. See Glycosides for more information on acidic hydrolysis of glycosides. Damascenone is also found in grapes and is a major flavor component of bourbon [22].
Other Flavor and Non-flavor Compounds
Several volatile carbonyl compounds are formed during beer aging. Acetaldehyde is one such major flavor compound formed during aging and one of the first documented in science, and is a result of ethanol being oxidized. The aldehyde trans-2-nonenal (threshold of 50-250 parts per trillion [22]) is derived from oxidized fatty acids. It is the compound responsible for the cardboard (also described as "lipstick") flavor in aged beers and was once thought of as the molecule responsible for beer staling. This interpretation was updated by studies by Van Eerde et al. and Narziss et al. where it was shown that trans-2-nonenal formation was dependent on temperature (a lot is formed at 40°C, but none is formed at 20°C), and other authors observed that trans-2-nonenal develops independently of dissolved oxygen. Carbonyl scavengers such as hydroxylamine might help to diminish the effects of carbonyl staling compounds such as trans-2-nonenal. Other aldehydes known as "strecker" aldehydes are formed during beer storage and increase depending on oxygen. While many of them are not impactful on flavor, their presence is indicative of an oxidation issue [18]. One such aldehyde that impacts the flavor of aging beer is the malt derived compound, methional (threshold of 4 ppb [22]). Methional is formed by the degradation of methionine by oxidation. The flavor of methional is described as "mashed potato" or "potato chips", and is common in wheat based beers [22][45].
Ketones are also formed during the storage of bottled beer. The buttery tasting compounds diacetyl and 2,3-pentanedione are examples of ketones formed during beer aging. These are more pronounced in beers that have higher dissolved oxygen during packaging. Diacetyl, in particular, can form levels that are above flavor threshold [18].
Many other compounds increase or diminish over time in packaged beer. Cyclic acetals (2,4,5-trimethyl-1,3-dioxolane, 2-isopro-pyl-4,5-dimethyl-1,3-dioxolane, 2-isobutyl-4,5-dimethyl-1,3-dioxolane and 2-sec-butyl-4,5-dimethyl-1,3-dioxo-lane) in packaged beer that has been in contact with oxygen. Heteroclyclic compounds (furan, furfural, and furanone based compounds) increase regardless of oxygen exposure but generally remain far below flavor threshold, however, their presence correlates with stale flavors in sensory tests and can thus serve as indicators for stale beer. Pyrazines such as pyrazine, 2-ethyl-6-methylpyrazine, 2-ethyl-5-methylpyrazine, 2-acetylpyrazine, 2,3-dimethylpyrazine, 2,5-dimethylpyrazine, 2-ethyl-3,6-dimethylpyrazine, 2-ethyl-3,5-dimethylpyrazine diminish over time in packaged beer [18].
Products of Maillard reactions, which include a diverse range of reactions, have also been found in beer, although research in this area is limited. Some Maillard compounds found in aging beer remain under taste threshold, for example, furfural and 5-hydroxymethyl furfural. It is hypothesized that a wide range of unknown Maillard reactions and their intermediates might play a role in the aging of beer. In particular, the bready, sweet, caramel and wine-like character of stale beer might be due to Maillard reactions [18].
In general, lower storage temperatures preserve hop compounds. Cans also help preserve some hop compounds versus bottles because bottle caps can strip certain hop compounds such as myrcene and caryophyllene when stored at room temperature (less so when stored cold). For example, one study found a moderate amount of degradation of humulinones, iso-α-acids, and residual α-acids when dry hopped beers were stored at 20°C versus 3°C. There was also an overall decrease in hop aroma compounds during warm storage, with some esters, hop monoterpenes, and sesquiterpenes showing poor storage stability compared to other ester compounds, monoterpene alcohols, and ketones which increased during warm storage. After 10 months of storage, the dry hopped beers stored at 20°C had a significant drop in floral, citrus and tropical fruit notes when compared to the same beers stored at 3°C [46].
Tetrahydropyridine (THP) is a compound that tastes like Cheerios® or corn tortilla chips that often develops soon after packaging beers that contain Brettanomyces or heterofermentative Lactobacillus. It is usually detected after swallowing the beer. This compound is stimulated by oxygen, and often ages out after a few months. See the Tetrahydropyridine page for more information.
Pediococcus 'sickness'
- See the Pediococcus page.
See Also
Additional Articles on MTF Wiki
MTF Facebook Discussions
- Devin Bell's facebook discussion about aging in bottles
- Discussion about how long it takes to develop carbonation
External Resources
References
- ↑ The influence of packaging material on volatile compounds of pale lager beer. Goran Gagula, Kristina Mastanjević, Krešimir Mastanjević, Vinko Krstanović, Daniela Horvat, Damir Magdić. 2020.
- ↑ CO2 volume fluxes outgassing from champagne glasses: The impact of champagne aging. Gérard Liger-Belair, Sandra Villaume, Clara Cilindre, Philippe Jeandet. 2010.
- ↑ 3.0 3.1 3.2 3.3 3.4 Losses of Dissolved CO2 Through the Cork Stopper during Champagne Aging: Toward a Multiparameter Modeling. Gérard Liger-Belair and Sandra Villaume. 2011.
- ↑ Kinetics of CO(2) fluxes outgassing from champagne glasses in tasting conditions: the role of temperature. Liger-Belair G1, Villaume S, Cilindre C, Jeandet P. 2009.
- ↑ Foaming properties of various Champagne wines depending on several parameters: Grape variety, aging, protein and CO2 content. Clara Cilindrea, Gérard Liger-Belair, Sandra Villaume, Philippe Jeandet, Richard Marchal. 2010.
- ↑ The impact of closure type and storage conditions on the composition, colour and flavour properties of a Riesling and a wooded Chardonnay wine during five years' storage. G.K.SKOUROUMOUNIS, M.J. KWIATKOWSKI, I.L. FRANCIS, H. OAKEY, D.L. CAPONE, B. DUNCAN, M.A. SEFTON, E.J. WATERS. 2005.
- ↑ 7.0 7.1 Ryan Fields and Jay Goodwin. The Brewing Network, The Sour Hour – Episode 87. 10/04/2018.
- ↑ Conversation with Pierre Tilquin on MTF regarding horizontal bottle storage. 01/08/2016.
- ↑ Wikipedia. Storage of Wine. Retrieve 04/14/2016.
- ↑ Conversation with Raf Soef on horizontal bottle storage for natural corks. 0414/2016.
- ↑ Caterer Search. "The Lay Down on Storage". 2001. Archive.org saved on 03/01/2012. Retrieved 02/02/2018.
- ↑ Dr. Miguel Cabral. "COMPOUNDS CALLED CORKLINS FOUND IN CORK-STOPPERED WINES". The Drinks Business website article. 06/07/2018. Retrieved 06/08/2018.
- ↑ Dr. Miquel Cabral. "STORING WINE ON ITS SIDE IS NONSENSE, SAYS SCIENTIST". The Drinks Business website article by Patrick Schmitt. 06/11/2018. retrieved 06/11/2018.
- ↑ Milk The Funk discussion on the humidity of the headspace within wine bottles. 06/11/2018.
- ↑ Conversation with Jeff Porn on horizontal corked bottle storage. 04/14/2016.
- ↑ Shawn Savuto. Milk The Funk Facebook group post on storing bottles sideways and THP development. 06/25/2018.
- ↑ Dalgliesh, C. E. (1977). Flavour stability. Proceedings of the European Brewery Convention Congress, 623–659.
- ↑ 18.00 18.01 18.02 18.03 18.04 18.05 18.06 18.07 18.08 18.09 18.10 18.11 The chemistry of beer aging – a critical review. Bart Vanderhaegen, Hedwig Neven, Hubert Verachtert, Guy Derdelinckx. 2006. DOI:https://doi.org/10.1016/j.foodchem.2005.01.006.
- ↑ Wikipedia. "Redox". Retrieved 09/03/2017.
- ↑ 20.0 20.1 20.2 20.3 20.4 20.5 20.6 Evaluating the Impact of Dissolved Oxygen and Aging on Dry-Hopped Aroma Stability in Beer. Bradley M. Barnette. Masters Thesis in Food Science and technology, Oregon State University. 2018.
- ↑ Decrease of Aged Beer Aroma by the Reducing Activity of Brewing Yeast. Daan Saison, David P. De Schutter, Nele Vanbeneden, Luk Daenen, Filip Delvaux and Freddy R. Delvaux. 2010.
- ↑ 22.0 22.1 22.2 22.3 22.4 22.5 Further Insights into the Role of Methional and Phenylacetaldehyde in Lager Beer Flavor Stability. M. Soares da Costa, C. Gonçalves, A. Ferreira, C. Ibsen, P. Guedes de Pinho, and A. C. Silva Ferreira. 2004.
- ↑ "HOW TO MEASURE DISSOLVED OXYGEN IN THE BREWERY". Hach Company pamphlet. Retrieved 10/23/2018.
- ↑ Free radical scavenging and antioxidant effects of lactate ion: an in vitro study. Groussard C, Morel I, Chevanne M, Monnier M, Cillard J, Delamarche A. 1985.
- ↑ Effect of lactic acid fermentation on antioxidant, texture, color and sensory properties of red and green smoothies. Raffaella Di Cagno, Giovanna Minervini, Carlo G. Rizzello, Maria De Angelis, Marco Gobbetti. 2011.
- ↑ Charles Bamforth and Aldo Lentini. Beer: A Quality Perspective. 2009. Pgs 85-109.
- ↑ Temperature Regulation PowerPoint. Cal Poly. Environmental Proteomics Laboratory.
- ↑ 28.0 28.1 28.2 "Measurements of Beer Collodial Stability" (Presentation). Laurent Mélotte. Sept 2008.
- ↑ 29.00 29.01 29.02 29.03 29.04 29.05 29.06 29.07 29.08 29.09 29.10 29.11 29.12 Structure, Organoleptic Properties, Quantification Methods, and Stability of Phenolic Compounds in Beer—A Review. D. Callemien and S. Collin. 2010. DOI: http://dx.doi.org/10.1080/87559120903157954.
- ↑ 30.0 30.1 Contribution of Hop Bitter Substances to Beer Staling Mechanisms. Williams and Wagner 1979.
- ↑ Phenols. Wikipedia website. Retrieved 04/17/2017.
- ↑ 32.0 32.1 Polyphenol. Wikipedia website. Retrieved 04/17/2017.
- ↑ 33.0 33.1 33.2 Guaiacol and 4-methylphenol as specific markers of torrefied malts. Fate of volatile phenols in special beers through aging. Scholtes C, Nizet S, Collin S. 2014.
- ↑ 34.0 34.1 The Effect of Hydroxycinnamic Acids and Volatile Phenols on Beer Quality. S. E. Iyuke, E. M. Madigoe, and R. Maponya. 2008.
- ↑ Flavonoid. Wikipedia website. Retrieved 05/02/2017.
- ↑ "Quinone". Wikipedia. Retrieved 12/23/2023.
- ↑ Tannic acid. Wikipedia website. Retrieved 04/17/2017.
- ↑ 38.0 38.1 Use of Tannic Acid in the Brewing Industry for Colloidal and Organoleptic Stability. Joseph A. Formanek and Pieter Bonte. MBAA Technical Quarterly. 2017.
- ↑ 39.0 39.1 Evaluation of the Addition of Gallotannins to the Brewing Liquor for the Improvement of the Flavor Stability of Beer. Guido Aerts, Luc De Cooman, Gert De Rouck, Zoltan Pénzes, Annemie De Buck, Roger Mussche, and Joseph van Waesberghe. 2004.
- ↑ "Episode 25 - BrewTan Your Questions Away". Experimental Brewing podcast (~25 minutes in). Retrieved 04/17/2017.
- ↑ A New Parameter for Determination of the Extent of Staling in Beer. Shigeki Araki, Masachika Takashio, and Ken Shinotsuka. 2002. DOI: 10.1094/ASBCJ-60-0026.
- ↑ 125th Anniversary Review: The Role of Hops in Brewing. Schönberger and Kostelecky. 2012. DOI: 10.1002/j.2050-0416.2011.tb00471.x.
- ↑ Dr. Patricia Aron. "Bitterness and the IBU: What’s It All About?" HomebrewCon 2017 Presentation. ~32 mins in. Retrieved 09/05/2017.
- ↑ Investigation of the β-Damascenone Level in Fresh and Aged Commercial Beers. Fabienne Chevance, Christine Guyot-Declerck, Jérôme Dupont, and Sonia Collin. 2002. DOI: 10.1021/jf020085i.
- ↑ Robert Hall, Andy Mitchell. Beer Oxidation: Chemistry, Sensory Evaluation, and Prevention. HomebrewCon 2017 Seminar. 2017.
- ↑ Kemp, O., Hofmann, S., Braumann, I., Jensen, S., Fenton, A., and Oladokun, O. (2021) Changes in key hop-derived compounds and their impact on perceived dry-hop flavour in beers after storage at cold and ambient temperature. J. Inst. Brew., https://doi.org/10.1002/jib.667.